J Biomed 2019; 4:35-43. doi:10.7150/jbm.33922 This volume Cite
Review
N-Glycosylation of IgG Immunoglobulin and its clinical significance
1. Blood Transfusion Service, General Hospital of Nikea, Andrea Petrou Mantouvalou, 184 54, Athens, Greece
2. Hematology Laboratory-Blood Bank, ARETAEIO Hospital , National and Kapodistrian University of Athens 76 Vasilissis Sofias Ave., 115 28, Athens, Greece
*These authors contributed equally.
Abstract
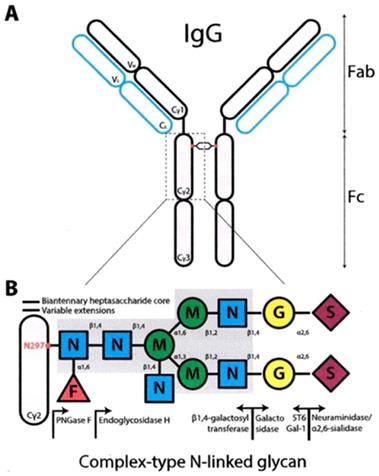
The IgG immunoglobulins are the main immunoglobulins in human beings. They have the longest half time and are the most studied of all. After the Ag binding, there is a signaling through the Fc region.
The post translational modification of the immunoglobulin mainly includes the N-Glycosylation and mostly the IgG one. The IgGs represent the antibodies, which are the mediators of the immunity, against extracellular bacteria and toxins.
The Fcγ receptors exist in all hemopeitic cells. The extracellular parts of the receptors show high grade of homology in their amino acid sequences. Monoclonal antibodies can distinguish them.
In addition to nucleic acids, proteins and lipids, sugars are also fundamental components of animal systems. Compared with advances in genomics and proteomics, the study of glycoscience is under investigation.
Changes in glycosylation may be used as biological markers for screening in relation to healthy subjects. It may help in the discovery of patients in early stages of the disease and thus be used as a biological marker to select patients with better prognosis and therefore to use as chemotherapy chemotherapeutic agents with lower toxicity.
Keywords: N-Glycosylation, Immunoglobulin